Introduction
Lipoedema and ‘GLP-1s’ are being discussed much more frequently, particularly on social media, where the language used is often inaccurate and, at times, misleading.
The term “GLP-1s” has become a catch-all, when in reality these medications sit within a broader group of incretin-based gut hormone therapies.
These therapies act on pathways involving glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP).
Medications now widely referred to as “GLP-1s” were originally developed for the management of type 2 diabetes, a condition of metabolic dysregulation. Their primary role was to improve glucose control through effects on insulin secretion, appetite signalling and energy balance. As their use has moved into weight management, so has the amount of misunderstanding and misinformation surrounding them.
What These Drugs Actually Are
It is important to be precise.
Semaglutide, used in Wegovy, is a GLP-1 receptor agonist, acting on that pathway.
Tirzepatide, used in Mounjaro, is a dual incretin agonist, acting on both GLP-1 and GIP pathways. GIP refers to glucose-dependent insulinotropic polypeptide, a second gut hormone involved in insulin secretion and metabolic regulation.
This distinction matters. These drugs are not acting through a single mechanism, and describing them all as “GLP-1s” is inaccurate.
The field is continuing to evolve. Newer agents such as Retatrutide are being developed as triple agonists, acting across GLP-1, GIP and glucagon pathways, specifically to target obesity instead of diabetes. This reflects a broader shift towards multi-pathway metabolic regulation rather than a single class of drugs.

Mounjaro, Inflammation and Wider Metabolic Effects
Beyond appetite regulation, incretin-based gut hormone therapies influence a broader set of physiological systems.
Tirzepatide (Mounjaro), as a dual incretin agonist, has effects that extend beyond glucose control and weight reduction. These include changes in insulin sensitivity, lipid metabolism and inflammatory signalling.
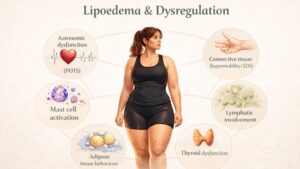
There is increasing evidence that these medications reduce markers of low-grade systemic inflammation. This is relevant in lipoedema, where inflammatory processes and altered tissue environments form part of the clinical picture.
Improved metabolic regulation may also influence vascular function and tissue perfusion. Whilst not a direct treatment for lipoedema tissue, it alters the physiological environment in which that tissue exists.
This helps explain why changes may be seen in symptoms such as heaviness, discomfort and fatigue, even where structural changes remain.
The key point is that these effects are systemic rather than local. The benefit, where it occurs, is through regulation of metabolism and inflammation rather than targeted modification of lipoedema fat.
Why These Medications Are Being Used in Lipoedema
The relevance of incretin-based therapies such as semaglutide and tirzepatide in lipoedema sits within their broader regulatory effects, rather than a single targeted mechanism.
Lipoedema is not simply a condition of fat accumulation. It involves altered adipose tissue behaviour, microvascular dysfunction and a background of chronic low-grade inflammation. Semaglutide and tirzepatide influence appetite regulation, metabolic signalling, insulin sensitivity and inflammatory pathways, all of which intersect with this clinical picture.
These medications are not a direct treatment for lipoedema itself, but their successful use has challenged the long-standing belief that lipoedema fat is resistant to change. In practice, improved metabolic regulation and weight reduction can influence symptom burden, tissue behaviour and overall progression.
This shifts the narrative. Weight management is not irrelevant in lipoedema – it is simply more complex.
By improving metabolic regulation and reducing systemic inflammatory load, these medications may reduce overall physiological stress. This can translate into changes in pain, heaviness, fatigue and function, even where tissue structure itself remains.
Food Noise and Appetite Regulation
One of the most consistent effects described is the reduction in what is often referred to as “food noise”.
This is the persistent background drive towards food, thinking about eating, planning meals or feeling compelled to eat without true physiological hunger. With incretin-based therapies, this signalling becomes quieter or stops altogether, appetite reduces, satiety increases and eating becomes more regulated rather than effort-driven.
This is not simply appetite suppression. It reflects a shift in central appetite regulation, reward pathways and energy balance.
How They Are Delivered
Incretin-based therapies are administered as injections, typically once weekly, using a pre-filled pen device.
They are given into the subcutaneous fat layer, usually in the abdomen, thigh or upper arm. This allows for slow, consistent absorption into the bloodstream. Although injected into fat, these medications do not act locally. Their effects are systemic, influencing hormonal and metabolic pathways across the body.
Inflammation and Systemic Regulation
There is increasing recognition that these medications influence inflammatory signalling more broadly.
Improvements in insulin sensitivity, vascular function and metabolic regulation may contribute to symptom change beyond weight alone. In the context of lipoedema, where inflammation and tissue sensitivity are often present, this wider regulatory effect is clinically relevant.
Clinical Context and Limitations
It is important to be clear about what these treatments do and do not do.
These medications do not remove lipoedema tissue, reverse established fibrosis or selectively alter fat distribution in affected areas. Any reduction in size tends to reflect overall weight change rather than targeted modification of lipoedema-specific tissue.
From a clinical perspective, these medications sit alongside, not instead of, appropriate assessment and management. Their role is to support regulation where metabolic, inflammatory and appetite-related factors are contributing to the overall presentation.
Summary
Incretin-based therapies, including semaglutide (Wegovy) and tirzepatide (Mounjaro), represent a shift in how metabolic regulation, appetite control and systemic inflammation are understood and managed.
Although commonly grouped under the term “GLP-1s”, these medications act across multiple gut hormone pathways and should not be considered a single class of drugs. Tirzepatide, as a dual incretin agonist, extends beyond the GLP-1 pathway, reflecting a broader move towards multi-pathway metabolic intervention.
In the context of lipoedema, these medications do not directly alter the structure of affected tissue. However, they influence key regulatory systems that are relevant to the condition, including appetite signalling, insulin sensitivity, vascular function and inflammatory processes.
This has important implications. The long-standing view that weight management does not affect lipoedema is overly simplistic. While tissue behaviour remains distinct, improvements in metabolic regulation and reductions in systemic load can influence symptom burden, functional capacity and overall disease experience.
These therapies should therefore be understood as part of a wider regulatory framework rather than a targeted treatment. Their role is to support systemic balance, not to replace clinical assessment or management of lipoedema itself.