Introduction
Mast cells are part of normal human physiology and are best known for their role in allergic reactions. They sit within connective and adipose tissues, particularly around blood vessels, lymphatic vessels and peripheral nerves, as well as in areas such as the skin, respiratory mucosa and gastrointestinal lining where potential toxins, irritants or antigens from outside the body first come into contact with tissue. Their role is protective, rapid, and primarily local. This article explains what mast cells are and how mast cell activation syndrome (MCAS) might relate to tissue behaviour seen in lipoedema.
Mast Cells: Structure & Function
Mast cells are immune cells derived from the bone marrow that mature within tissues. Unlike circulating immune cells, they remain resident in place, acting as local sentinels.
They contain intracellular granules packed with pre-formed mediators, including histamine, proteases (such as tryptase), and other signalling molecules. When activated, they can release these substances rapidly (degranulation), and also generate additional mediators over time for sustained reactions, including prostaglandins, leukotrienes, and cytokines.
Their core functions are:
- Immediate defence against perceived threats (Allergic reactions)
- Regulation of vascular permeability
- Interaction with sensory nerves (including pain signalling)
- Participation in wound healing and tissue remodelling
In normal physiology, this system is tightly controlled. Activation occurs when appropriate, is proportionate, and resolves once the stimulus is removed.
Normal Mast Cell Responses
The clearest example of normal mast cell activation is an acute allergic or irritant response, such as a bee sting.
At the site of injury, mast cells release mediators including histamine. This causes local blood vessels to dilate and become more permeable, allowing fluid to move into the surrounding tissue. The result is swelling and redness. At the same time, nearby sensory nerves are activated, producing itching or pain.
When mediator release extends beyond a single localised site, these vascular effects can influence the circulation more broadly. Vasodilation and increased vascular permeability can reduce effective circulating volume, producing transient lightheadedness, flushing and a compensatory increase in heart rate.
This is a local response. It is proportionate to the stimulus and resolves once the stimulus has passed.
The same mechanism operates in other tissues in response to specific exposures:
- In the skin, mast cells activate in response to physical irritation, mechanical pressure, insect bites, or contact with irritants or allergens, producing a local wheal and flare response.
- In the airways, mast cells activate in response to inhaled particles such as pollen, dust or other environmental exposures, leading to temporary airway narrowing and increased mucus production.
- In the gastrointestinal tract, mast cells activate in response to ingested substances, including food components or microbial products. This can produce abdominal cramping, urgency, and diarrhoea through effects on gut muscle contraction and fluid secretion.
- In the cardiovascular system, mast cell mediators can influence vascular tone and circulating volume, producing transient flushing, lightheadedness, and a short-lived increase in heart rate.
In each case, the response is driven by exposure within that tissue. The mechanism is the same: local mediator release affecting blood vessels, fluid movement and nerve activity. The defining feature is that the response is controlled and self-limiting.
Mast Cell Activation Syndrome (MCAS)
MCAS describes dysregulated mast cell behaviour. The cells are normal. The change lies in their threshold for activation, the amount of mediator released, and the ability of the response to resolve.
Mast cells may activate in response to stimuli that would normally be tolerated, respond disproportionately, or fail to settle once activated.
Rather than resolving, the effects of mediator release may persist or recur, maintaining increased vascular permeability, ongoing fluid shift, and sustained nerve sensitisation. This contrasts with normal mast cell responses, where activation is brief, proportionate, and self-limiting.

Clinical Presentation of MCAS
Persistent increases in vascular permeability allow continued movement of fluid into the interstitial space. This increases demand on lymphatic drainage and may present as ongoing or fluctuating swelling.
At the same time, continued exposure of peripheral nerves to mast cell mediators lowers activation thresholds. Sensory input is amplified, so that stimuli such as pressure or contact may become uncomfortable, painful, or intolerable.
Because mast cells are distributed across multiple tissues, these effects are not confined to a single system. They represent the same underlying process occurring in different anatomical sites.
Clinically, mast cell activation does not present as a single, isolated symptom. It more often appears as a pattern of seemingly unconnected inflammatory and vascular responses affecting multiple systems.
Common features include:
- Flushing or sudden warmth
- Itching, urticaria or skin reactivity
- Swelling or a sense of fluid fluctuation
- Lightheadedness or presyncope
- Tachycardia or palpitations
- Abdominal pain, bloating, diarrhoea or urgency
- Nausea
- Headache
- Sensory hypersensitivity, including pain to light touch
- Respiratory symptoms such as throat tightness or wheeze
These symptoms may occur together or in varying combinations, and may fluctuate over time. The unifying feature is not the specific symptom, but the pattern of mediator-driven effects across different tissues.
IgE-Mediated & Non-IgE Mast Cell Activation
Mast cells can be activated through different pathways. In classical allergic reactions, activation occurs through an IgE-mediated mechanism. IgE antibodies are produced in response to a specific allergen and bind to receptors on the surface of mast cells. When that allergen is encountered again, it cross-links these IgE molecules and triggers mast cell activation.
This leads to rapid release of mediators including histamine, tryptase, prostaglandins and leukotrienes. The effects are immediate and reproducible, driven by a specific allergen.
Mast cells can also be activated without IgE. They respond to physical triggers such as pressure or temperature change, chemical exposures, infections, medications, and signals from the nervous system. These pathways do not depend on allergen-specific IgE.
In these cases, the same mediators are released and the same tissue effects occur, but the trigger is not linked to a single identifiable allergen and may not be consistent.
Testing For MCAS
Standard allergy testing looks for IgE-mediated responses to specific allergens. This relies on a consistent relationship between an allergen and mast cell activation. Where a person’s symptoms follow that pattern, skin prick testing or serum-specific IgE testing can identify sensitisation to a defined trigger.
Where mast cell activation occurs through non-IgE pathways, these tests will be negative. This does not exclude mast cell mediator release. It reflects a different mechanism.
This is why many people present with obvious symptoms of MCAS but have normal allergy testing.
Biochemical testing is aimed at detecting mediator release rather than identifying a trigger. Serum tryptase is the most commonly used marker. It is most useful when measured during an acute episode and compared with a baseline level taken when the patient is well. A rise from baseline supports mast cell activation. A single normal result does not exclude it.
Other markers, including urinary histamine metabolites, prostaglandins and leukotrienes, may be measured in some laboratories. These are more variable, dependent on collection and timing, and less consistently available. They may support a pattern but do not define it in isolation.
Panels that claim to identify sensitivity to multiple foods, environmental exposures or chemicals do not establish mast cell dysregulation. They do not demonstrate how mast cells are behaving within tissue and do not distinguish between IgE-mediated allergy and non-specific reactivity.
Testing therefore has limitations. Results need to be interpreted alongside the clinical picture, with attention to timing, pattern of symptoms and response over time, rather than used as a single point of confirmation.
What MCAS Is Not
Mast cells are present in all connective tissues. Their presence is normal and does not indicate disease. MCAS is not the same as classical allergy, and it is not defined by a single laboratory test. It is not a uniform condition with a single cause.
MCAS is not established by identifying mast cells within tissue, nor by broad panels suggesting multiple sensitivities without a defined mechanism.
It represents a pattern of dysregulated mediator release, identified through consistent clinical features rather than isolated findings.
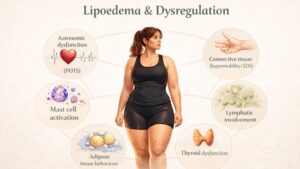
Lipoedema & MCAS
Mast cells are present within adipose tissue as part of normal connective tissue. They form part of the stromal environment within fat. Mast cells sit within adipose tissue and act locally on vascular permeability, fluid movement and nerve signalling. In lipoedema, where adipose tissue behaviour is altered, this provides a direct route by which mast cell activity could influence hypersensitivity, fluid shift and tissue reactivity.
In lipoedema, attention has been drawn to these cells because of the clinical pattern seen. Features such as marked hypersensitivity within the skin and subcutaneous tissue, pain to light touch or pressure, and a sense of tissue reactivity are not fully explained by fat distribution alone.
Flushing, itching, and exaggerated responses to minor stimuli may also be present. In some cases, gastrointestinal symptoms sit alongside these features.
Because mast cells influence vascular permeability, fluid movement and nerve signalling, they have been considered as a possible contributor to this pattern.
Research has examined whether mast cell number, distribution or activity within adipose tissue differs in lipoedema, and whether this could relate to inflammation, pain or tissue sensitivity.
Findings are variable and do not establish a consistent mechanism. There is no clear evidence that mast cell dysregulation defines lipoedema or explains it as a condition.
Mast Cell Mechanisms Within Lipoedema Tissue
There is evidence of low-level inflammatory activity within lipoedema tissue. Within that environment, mast cell mediator release provides a plausible mechanism for how local tissue behaviour becomes amplified.
Histamine, prostaglandins and leukotrienes increase vascular permeability, allowing repeated movement of fluid into the interstitial space. This is not a single, resolving shift, but a process that can continue, increasing tissue load and tension.
At the same time, these mediators act directly on peripheral nerves, lowering activation thresholds and amplifying sensory input. This provides a mechanism for hypersensitivity, including pain to light touch and exaggerated responses to pressure.
Taken together, this links fluid dynamics, inflammation and nerve sensitivity within the same tissue environment.
This does not redefine lipoedema as a mast cell disorder. It describes how mast cell activity, within an already altered adipose environment, could contribute to the local tissue features observed.
Interpreting Overlap with Lipoedema
Symptoms such as hypersensitivity, flushing, gastrointestinal disturbance, and variability across systems are often grouped together and attributed to mast cell activation.
These features are not specific. Within lipoedema, local tissue behaviour can account for many of these presentations. Altered adipose tissue, increased interstitial fluid, low-level inflammatory activity, and changes in nerve sensitivity can produce pain, pressure sensitivity, and a sense of tissue reactivity.
Mast cells act on the same processes. Their mediators influence vascular permeability, fluid movement, inflammatory signalling and nerve activation. When these effects are sustained within tissue, the clinical picture can resemble mast cell mediator-driven symptoms.
This creates a point of overlap. The distinction lies in pattern. MCAS is defined by recurrent, multi-system episodes of mediator release. Local tissue changes within lipoedema do not require systemic mast cell activation to explain them.
For this reason, there is a risk of misattribution. Non-specific symptoms may be labelled as mast cell activation without a clear mechanism or pattern to support that diagnosis.
At the same time, the overlap is clinically persuasive because the same physiological processes are involved. Dysregulation of vascular permeability, interstitial fluid dynamics, inflammatory signalling and nerve sensitivity can produce similar features, even when the underlying drivers differ.
This places the current understanding at the level of plausible mechanism. Mast cell activity within adipose tissue may contribute to the local tissue environment in lipoedema, particularly where hypersensitivity and reactivity are prominent.
It does not establish MCAS as a defining feature or cause. Clinical assessment therefore requires separation of these patterns. Systemic, episodic, mediator-driven symptoms should be considered on their own basis. Localised tissue behaviour should be interpreted within the context of the tissue itself.
Conclusion
Mast cells are a normal part of connective tissue, positioned to regulate vascular permeability, fluid movement and nerve signalling in response to environmental exposure.
MCAS reflects dysregulation of this system, with mediator release that is excessive, poorly controlled or inappropriately triggered, affecting multiple systems.
Within lipoedema, mast cells form part of the adipose tissue environment. Their presence is expected. The interest in them arises from the clinical features seen within the tissue, particularly hypersensitivity, fluid fluctuation and low-level inflammation.
There is a clear overlap in mechanism. Both mast cell activity and adipose tissue behaviour influence the same local processes, which provides a framework for understanding how these features can arise within lipoedema without requiring a single unifying diagnosis.
At present, this remains an area of investigation. Mast cell activity may contribute to the local tissue environment but it does not define lipoedema as a mast cell disorder.
The clinical task is to distinguish between systemic patterns of mast cell activation and local tissue behaviour, recognising where they may overlap, without conflating the two.
Bibliograpy
Valent P, Akin C, Bonadonna P, Hartmann K, Brockow K, Niedoszytko M, et al. Proposed diagnostic algorithm for patients with suspected mast cell activation syndrome. J Allergy Clin Immunol Pract. 2019;7(4):1125–1133.e1.
Weiler CR, Austen KF, Akin C, Barkoff MS, Bernstein JA, Bonadonna P, et al. AAAAI mast cell disorders committee work group report: mast cell activation syndrome (MCAS) diagnosis and management. J Allergy Clin Immunol. 2019;144(4):883–896.
Gülen T, Hägglund H. Selecting the right criteria and proper classification to diagnose mast cell activation syndromes: a critical review. J Allergy Clin Immunol Pract. 2021;9(10):3918–3928.
Akin C, Valent P, Metcalfe DD. Mast cell activation syndrome: proposed diagnostic criteria. J Allergy Clin Immunol. 2010;126(6):1099–1104.e4.
Divoux A, Clément K. Architecture and the extracellular matrix: the still unappreciated components of adipose tissue. Obes Rev. 2011;12(5):e494–e503.
Poggi M, Jager J, Paulmyer-Lacroix O, et al. Activation of adipose tissue mast cells in human obesity. Diabetes. 2007;56(5):1357–1365.
Liu J, Divoux A, Sun J, et al. Genetic deficiency and pharmacological stabilisation of mast cells reduce diet-induced obesity and diabetes in mice. Nat Med. 2009;15(8):940–945.
Al-Ghadban S, Cromer W, Allen M, Ussery C, Badowski M, Harris D, et al. Dilated blood and lymphatic microvessels, angiogenesis, increased macrophages, and adipocyte hypertrophy in lipoedema thigh skin and fat tissue. J Obes. 2019;2019:8747461.
Czerny M, Schmeller W, et al. Lipoedema: a call to action. J Eur Acad Dermatol Venereol. 2020;34(10):e537–e539.
Wold LE, Hines EA, Allen EV. Lipedema of the legs: a syndrome characterised by fat legs and oedema. Ann Intern Med. 1951;34(5):1243–1250.
Herbst KL. Rare adipose disorders (RADs) masquerading as obesity. Acta Pharmacol Sin. 2012;33(2):155–172.