Introduction
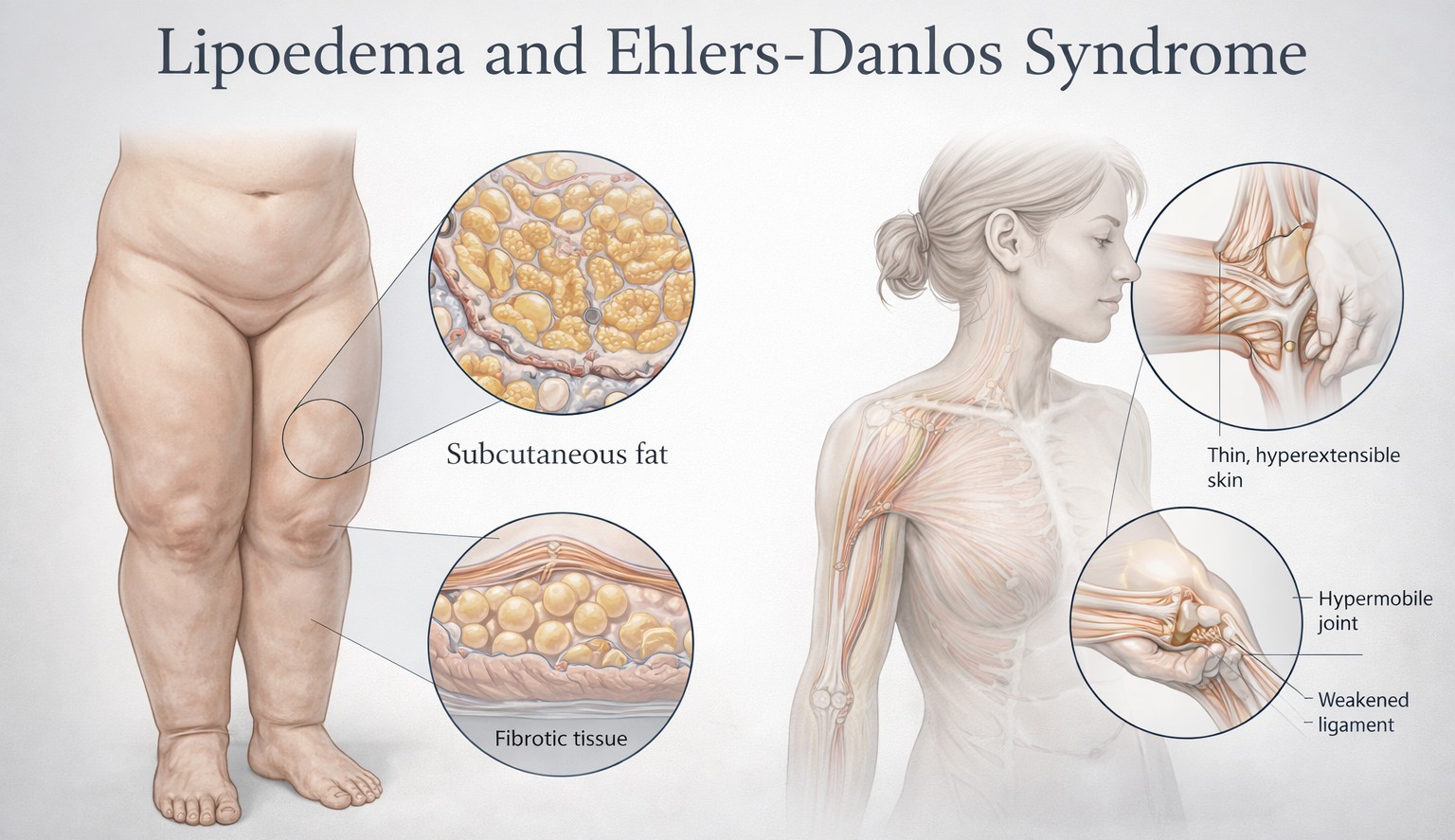
Lipoedema is progressively being understood not simply as a disease of fat distribution, but in many women, as a condition involving loose connective tissue, fascia, microvasculature, extracellular matrix remodelling and hypermobility.
This matters because many women with lipoedema describe symptoms that do not fit neatly into a “fat disorder” model alone: easy bruising, pain, tissue softness, joint laxity, venous problems, prolapse, hernias, pelvic symptoms, poor tissue support, autonomic complaints and a strong family history suggest broader connective tissue dysfunction.
The most recognised connective tissue disorder relating to lipoedema is hypermobile Ehlers-Danlos syndrome, often abbreviated to hEDS, together with associated Hypermobility Spectrum Disorders, or HSD. These are not the same diagnosis as lipoedema, but the overlap of symptoms is clinically important. Joint hypermobility has been reported in a substantial proportion of women with lipoedema, and recent studies have argued that connective tissue laxity may contribute to the vascular, lymphatic and musculoskeletal features seen in both conditions.
What is Connective Tissue?
Connective tissue is the body’s supporting framework that gives strength, stretch, recoil and resilience to bodily structures. When connective tissue is unusually lax, fragile or disorganised, the effects can be widespread: tendons do not support joints, so they move beyond their normal range or sit incorrectly within joint capsules, veins bulge and distend, tissue bruises more easily and fasciae give less support so spinal discs, organs or pelvic structures are more vulnerable to prolapse or mechanical strain.
In lipoedema, connective tissue is significant because fat is not biologically separate from the surrounding connective tissue. Adipose (fat) tissue is supported by septa, fascia, microvessels, immune cells and extracellular matrix. If these structures are altered, the tissue can become mechanically different: softer in some areas, fibrotic in others, more painful, more fluid-laden and less capable of normal recoil. That is one reason many experts no longer describe lipoedema as “just fat”.
What is the Extracellular Matrix?
The extracellular matrix, or ECM, is the scaffold surrounding cells. It is made up of collagen, elastin, and ground substance made of glycoproteins, proteoglycans and molecules such as hyaluronan. In practical terms, it is the material that helps tissues hold their shape, transmit force, bind water, regulate cell signalling and respond to injury. In adipose tissue, the ECM determines how fat cells expand, how much fibrosis develops, how fluid moves through tissue and how inflammation alters tissue architecture.
This is highly relevant to lipoedema. Clinical reviews describe altered ECM states in lipoedema, including loss of elastic recoil, increased interstitial fluid, fibrosis and abnormal remodelling of adipose connective tissue. Hyaluronan has become a particular focus because it binds water, affects tissue hydration and may contribute to adipose expansion, inflammation and collagen deposition. This helps explain why lipoedema tissue can feel both soft and heavy, yet also nodular, fibrotic or mechanically abnormal.
What is Ehlers-Danlos Syndrome?
The NHS describes Ehlers-Danlos syndromes as inherited conditions affecting connective tissue that supports skin, tendons, ligaments, blood vessels, internal organs and bones.
In 2017, the International EDS Consortium convened to revise the diagnostic framework for the Ehlers-Danlos syndromes. The resulting International Classification recognises 13 types of EDS, although older descriptions and informal discussions may refer more loosely to a larger number of variants and related syndromes. The best-known forms include classical EDS, vascular EDS and hypermobile EDS.
Hypermobility Spectrum Disorders
Not everyone with symptomatic hypermobility has hypermobile EDS, so in 2017, the consortium also formalised hypermobility spectrum disorders (HSD) for individuals with symptomatic joint hypermobility who do not meet the full criteria for hypermobile EDS or another heritable connective tissue disorder. Clinically, many individuals with HSD may be as significantly affected as those with hypermobile EDS.
Hypermobile EDS
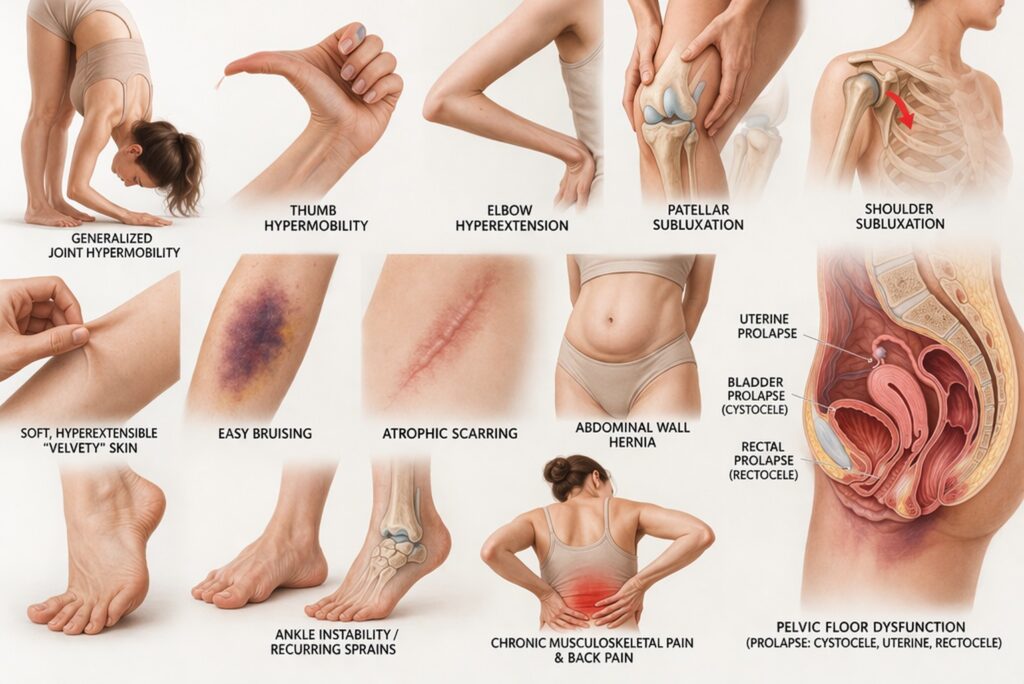
Hypermobile EDS is the subtype most often discussed in relation to lipoedema. It is characterised by generalised joint hypermobility, joint instability, chronic musculoskeletal pain and a broader pattern of connective tissue symptoms such as soft, velvety or hyperextensible skin, easy bruising, hernias, prolapse and tissue fragility.
Importantly, unlike several other EDS subtypes, hypermobile EDS currently has no confirmed single causative gene for routine clinical testing, so, like lipoedema, diagnosis remains clinical rather than molecular.
Why is hypermobility relevant for lipoedema?
Several lipoedema papers and reviews note that hypermobile joints are frequent in affected women, so the overlap is too common to ignore. Some studies have reported rates around half, and some even higher, although exact percentages vary according to methods and case selection. Researchers have suggested that this may reflect altered connective tissue compliance, loss of tissue elasticity and increased vessel distensibility, which in turn could affect capillary leakage, interstitial fluid dynamics, venous function and pain.
This does not mean that lipoedema is simply “undiagnosed EDS”, nor that every woman with lipoedema has a connective tissue syndrome. It means that in a meaningful subgroup, lipoedema appears to sit within a broader phenotype of connective tissue vulnerability.
That broader phenotype may include hypermobility, chronic pain, venous insufficiency, pelvic floor weakness, autonomic symptoms, gastrointestinal symptoms and family clustering of similar traits. Recent observational work has argued that lipoedema and HSD may share pathophysiological mechanisms, though this remains an emerging research area.
How Might the Two Conditions Connect Biologically?
Several mechanisms are plausible.
- Collagen and matrix abnormalities may reduce tissue recoil and structural support. In practical terms, looser tissue may permit greater distension of vessels and greater interstitial fluid movement. Lipoedema studies have proposed exactly this: that altered connective tissue structure may allow more fluid to leave the microvasculature and accumulate in the interstitium.
- Fascia and adipose tissue are mechanically linked. If fascial support is abnormal, load distribution through the limbs changes. This may contribute to pain, gait change, knee strain and a sense of heaviness or instability.
- Extracellular Matrix remodelling appears central in lipoedema. Increased hyaluronan, fibrosis and adipose tissue remodelling may alter both biomechanics and local inflammation.
- Vascular and lymphatic behaviour may be affected by connective tissue properties. Veins and lymphatic vessels rely on surrounding tissue support as well as intrinsic wall structure. If connective tissue is lax or mechanically abnormal, this may contribute to venous dilation, easy bruising, fluid accumulation and reduced efficiency of tissue clearance
- Genetics may overlap at least in part. Family-based genetic work in lipoedema has identified ECM-related rare variants, including discussion of tenascin-X pathways relevant to connective tissue biology. This does not prove that lipoedema is an EDS subtype, but it strengthens the argument that matrix biology belongs at the centre of lipoedema research.
What Does hypermobile EDS Actually Look Like in Clinic?
Patients with hEDS often describe being “double-jointed” since childhood, frequent sprains, recurrent subluxations or dislocations, chronic pain, fatigue, poor proprioception, pelvic instability, easy bruising, gastrointestinal symptoms and difficulty tolerating prolonged standing or repetitive load. Skin may feel soft or velvety, stretch may be mildly increased and scars may not behave normally, though the dramatic skin signs of classical EDS are often absent. Hernias, disc and organ prolapses and varicose veins may occur.
In a woman with lipoedema, these signs can easily be missed because clinicians may focus only on body shape. But if she also reports ankle instability, knee hyperextension, recurrent shoulder issues, pelvic girdle pain in pregnancy, hernias, prolapse or a family history of “loose joints”, prolapse, or hernias, then the possibility of hEDS or HSD becomes clinically relevant. Core sources for this phenotype are GeneReviews and the 2017 diagnostic framework.
Lipoedema as a Connective Tissue Disorder
A growing number of publications explicitly frame lipoedema as a connective tissue disorder, or at least as a disorder of loose connective tissue and adipo-fascial tissue. This language is not merely semantic. It changes how the condition is being conceptualised. It helps explain why manual therapies, compression, movement strategies, posture, pain education, muscle support and connective tissue-aware rehabilitation may matter alongside weight-neutral metabolic care and, in selected cases, surgery.
It also helps explain why women with lipoedema often say, correctly, that the condition feels systemic. The tissue is painful, bruises easily, behaves differently under load and may worsen at hormonal transition points such as puberty, pregnancy and menopause. Hormonal reviews continue to support a role for female sex hormones in triggering or amplifying lipoedema expression, even though the exact endocrine mechanism is not yet settled.
Pregnancy, Hormones and Miscarriage
Pregnancy is important in both lipoedema and hypermobility disorders. Many women with lipoedema report onset or worsening at puberty, pregnancy or menopause, supporting the view that hormonal shifts influence tissue behaviour. Reviews increasingly discuss oestrogen, progesterone and connective tissue signalling as relevant to lipoedema biology, though the evidence remains incomplete.
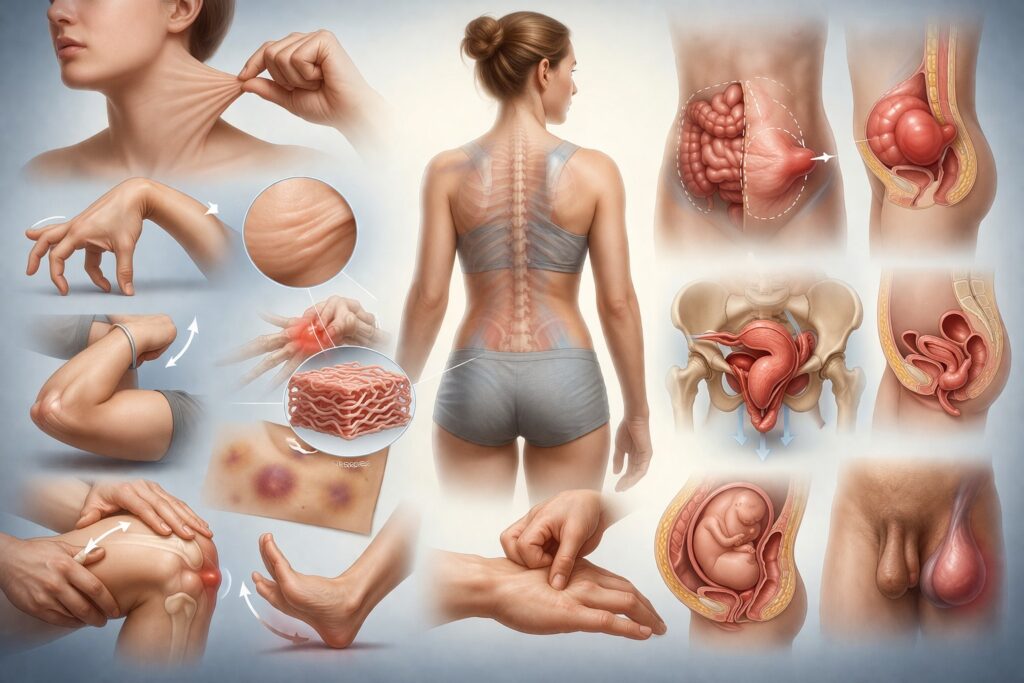
In hEDS and HSD, pregnancy can be physically demanding because lax tissues may become more symptomatic under the effects of hormonal softening, increased blood volume and altered biomechanics. Contemporary guidance notes issues such as pelvic girdle pain, joint instability, fatigue, dysautonomia, gastrointestinal symptoms, pubic symphysis syndrome, increased or prolonged bleeding in some patients, sometimes leading to placental abruption and late miscarriage.
Miscarriage needs careful wording. It would be inaccurate to say that everyone with hEDS has a high miscarriage risk. The most up-to-date expert guidance reports that overall miscarriage risk in hEDS/HSD is not clearly higher than in the general population, but recurrent miscarriage appears to be increased in a subgroup. Other studies, including survey and registry work, have reported higher rates of adverse pregnancy outcomes in vascular EDS populations overall, though this literature includes different EDS subtypes and methods, so it should not be oversimplified.
That distinction is important for patients. A history of pregnancy loss should be taken seriously, but it should not automatically be attributed to hypermobility. Nor should hypermobility be dismissed as irrelevant. The right conclusion is that connective tissue disorders can affect reproductive tissues, cervical competence, bleeding, pelvic support and mechanical symptoms in pregnancy, but the exact risk profile depends on subtype and individual history. Vascular EDS is a different and much higher-risk condition, and it should never be conflated with hEDS.
Pelvic Floor, Prolapse and Gynaecological Symptoms
One of the most practically important areas of overlap is pelvic support. Connective tissue laxity may predispose to pelvic floor dysfunction, prolapse, stress urinary symptoms and difficult postpartum recovery. These are well recognised in hEDS and often described by women with lipoedema and hypermobility traits. If a patient has lipoedema plus chronic pelvic symptoms, this should not be written off as incidental. It may reflect a broader connective tissue phenotype requiring integrated assessment.
The Autism Connection: What Can Honestly Be Said?
This area needs care. There is a growing body of literature suggesting an association between autism, joint hypermobility, HSD and hEDS. Studies have found elevated rates of autistic traits, ADHD and autism diagnoses in hypermobile populations, and vice versa. A 2025 systematic review and meta-analysis concluded that the available evidence suggests an association, although the studies are heterogeneous and the field is still developing.
There are also papers suggesting that mothers with EDS/HSD report autistic children at relatively high rates. However, this remains an emerging research signal, not a settled causal pathway. It should not be presented as “women with EDS have autistic children because of their connective tissue disorder”.
The evidence supports an association worth further study, not a definitive explanation. Potential shared mechanisms discussed in the literature include genetics, neurodevelopment, autonomic dysfunction, immune dysregulation and extracellular matrix biology, but these remain hypotheses rather than proven causal chains.
For patient education, the most honest wording is this: hypermobility, hEDS/HSD and autism appear to overlap more often than expected by chance in some studies, and some families report both.
That does not mean one straightforwardly causes the other, and it does not mean every hypermobile mother will have an autistic child. It does mean clinicians should be alert to neurodivergence, sensory sensitivity, chronic pain and autonomic vulnerability when assessing hypermobile patients.
Why the Overlap is Often Missed in Women
Women with lipoedema and hypermobility are frequently mislabelled. Pain may be attributed to weight, bruising to “clumsiness”, pelvic symptoms to childbirth alone, fatigue to anxiety, and hypermobility to being “naturally flexible”. hEDS itself is often diagnosed late because there is no confirmatory blood test for routine use and because clinicians may only think of rare dramatic EDS phenotypes rather than common hypermobile presentations. Reviews continue to describe HSD and hEDS as under-recognised, multi-system conditions.
In lipoedema, that diagnostic delay is compounded by poor awareness and by persistent confusion with obesity, lymphoedema or chronic venous disease. The result is that women with a combined lipoedema-hypermobility phenotype may spend years without anyone joining the dots.
What Should Clinicians Look For?
Clinicians assessing lipoedema should think beyond limb shape. Relevant questions include childhood flexibility, recurrent sprains, dislocations or subluxations, pelvic girdle symptoms, easy bruising, soft or stretchy skin, hernias, prolapse, varicose veins, fatigue, dysautonomia, gastrointestinal complaints, chronic widespread pain and family history of hypermobility or connective tissue problems.
A Beighton score may be useful, but it is not the whole story, particularly in adults whose flexibility has reduced with age, pain or deconditioning. The 2017 hEDS criteria allow historical features to support assessment when the current Beighton score is borderline.
A downloadable evidence-based diagnostic criteria PDF for hEDS is available here
Patients with clear red flags for rarer monogenic EDS types, especially vascular or classical features, require referral to specialist genetics services.
What does this Mean for Management?
Recognising connective tissue involvement does not change the core fact that lipoedema has no single universal cure, but it can improve management significantly by:
- Supporting a weight-neutral, tissue-aware approach rather than repeated moralising about diet.
- Justifying attention to pain, orthopaedic load, gait, muscular support, proprioception and pacing.
- Recognising careful use of compression, especially where venous or lymphatic symptoms coexist.
- Explaining why some patients need pelvic floor input, brace support, strength-focused physiotherapy or autonomic management.
- Helping to frame surgery realistically: surgery may reduce pathological tissue burden in selected patients, but it does not “cure” an underlying connective tissue phenotype.
For patients, this recognition is often validating. Many have sensed for years that their issue is not simply body fat. The connective tissue framework explains why their tissues bruise, ache, swell, move and heal differently.
What Remains Uncertain?
A great deal. Lipoedema research is expanding, but biological certainty is still lacking. There is not yet proof that most lipoedema is a formal hereditary connective tissue disorder in the same sense as EDS. There is not yet proof of one unifying ECM defect, one hormonal mechanism or one genetic explanation. Likewise, the autism-hypermobility literature is promising but not definitive, and pregnancy outcome data vary across studies and EDS subtypes.
Therefore, the most accurate modern position is that lipoedema shows substantial evidence of connective tissue involvement, hypermobility is common in lipoedema, hEDS/HSD frequently overlap with it, and extracellular matrix biology is likely central to at least part of the disease process. But the field is still moving from clinical observation toward physiological proof.
Conclusion
Lipoedema should no longer be framed as a purely adipose tissue problem. For many patients, it is better understood as a disorder of adipose and loose connective tissue with important implications for pain, biomechanics, vascular permeability, lymphatic behaviour and hormonal transitions. Hypermobile Ehlers-Danlos syndrome and hypermobility spectrum disorders are not synonymous with lipoedema, but they represent one of the most important overlapping clinical patterns.
That overlap helps explain why some women with lipoedema also experience joint instability, bruising, pelvic floor dysfunction, difficult pregnancies, chronic pain and a wider family history of connective tissue fragility. It also explains why extracellular matrix biology has become such a serious area of research.
For patients, the key message is that these symptoms belong together more often than many clinicians realise.
For clinicians, the message is that lipoedema assessment should include connective tissue assessment, especially hypermobility, tissue fragility and multisystem features.
Bibliography and Recommended Reading
Core lipoedema papers and reviews
- Allen M, Schwartz RS, Herbst KL. Interstitial fluid in lipedema and control skin. Lymphat Res Biol. 2020;18(5):435–443.
- Cifarelli V, et al. Lipedema: progress, challenges, and the road ahead. Front Endocrinol (Lausanne). 2025.
- Duhon BH, Bartholomew JR, Herbst KL. Current mechanistic understandings of lymphedema and lipedema: tales of fluid, fat and fibrosis. Int J Mol Sci. 2022;23(9):4576.
- Herbst KL, Kahn LA, Iker E, et al. Standard of care for lipedema in the United States. Phlebology. 2021;36(10):779–796.
- Poojari A, Zheng G, Yin H, et al. Lipedema: insights into morphology, pathophysiology, diagnosis and treatment strategies. Biomedicines. 2022;10(11):2982.
- Rabiee A, et al. Lipedema and adipose tissue: current understanding, challenges, and future directions. Biomedicines. 2025.
- Sanchez-De la Torre Y, Wadeea R, Rosas V, Herbst KL. Lipedema: friend and foe. Int J Mol Sci. 2018;19(10):2874.
Lipoedema and hypermobility / connective tissue overlap
- Fiengo E, Sbarbati A. Lipedema and hypermobility spectrum disorders sharing pathophysiology: a cross-sectional observational study. J Clin Med. 2025.
- Morgan S, Jones RL, Fennell J, et al. A family-based study of inherited genetic risk in lipedema. Lymphat Res Biol. 2024.
- Rasmussen JC, Aldrich MB, Fife CE, et al. Lymphatic function and anatomy in early stages of lipedema. Obesity (Silver Spring). 2022;30(4):885–892.
- Wright TF, Herbst KL. A young woman with excessive fat in lower extremities develops disordered eating and is subsequently diagnosed with anorexia nervosa, lipedema and hypermobile Ehlers–Danlos syndrome. Am J Case Rep. 2021;22:e931978.
Ehlers-Danlos Syndrome and Hypermobility
- Carroll MB. Hypermobility spectrum disorders: a review. Rheumatol Immunol Res. 2023;4(3):134–140.
- Castori M, Tinkle B, Levy H, et al. A framework for the classification of joint hypermobility and related conditions. Am J Med Genet C Semin Med Genet. 2017;175(1):148–157.
- Hakim A, O’Callaghan C, De Wandele I, et al. Hypermobile Ehlers–Danlos syndrome. In: GeneReviews®. Seattle (WA): University of Washington; updated 2024.
- Malfait F, Francomano C, Byers P, et al. The 2017 international classification of the Ehlers–Danlos syndromes. Am J Med Genet C Semin Med Genet. 2017;175(1):8–26.
- Morlino S, Castori M. Placing joint hypermobility in context: traits, disorders and syndromes. Br Med Bull. 2023;146(1):3–18.
- NHS. Ehlers-Danlos syndromes. Available from: https://www.nhs.uk/conditions/ehlers-danlos-syndromes/
- NHS. Joint hypermobility syndrome. Available from: https://www.nhs.uk/conditions/joint-hypermobility-syndrome/
Pregnancy, Miscarriage and Pelvic issues in hEDS/HSD
- Haem T, et al. Vascular Ehlers–Danlos syndrome and pregnancy: a systematic review. BJOG. 2024.
- Kang J, et al. Ehlers–Danlos syndrome in pregnancy: a review. Eur J Obstet Gynecol Reprod Biol. 2020;253:78–83.
- Pearce G, et al. Childbearing with hypermobile Ehlers–Danlos syndrome and hypermobility spectrum disorders: a large international survey of outcomes and complications. Int J Environ Res Public Health. 2023;20(20):6939.
- Pezaro S, et al. Management of childbearing with hypermobile Ehlers–Danlos syndrome and hypermobility spectrum disorders: a scoping review and expert co-creation of evidence-based clinical guidelines. PLoS One. 2024;19(5):e0301235.
- Spiegel E, et al. Pregnancy outcomes in women with Ehlers–Danlos syndrome. J Matern Fetal Neonatal Med. 2022;35(25):9641–9647.
Extracellular Matrix, Adipose Tissue and Fibrosis
- de Sousa Neto IV, et al. Adipose tissue extracellular matrix remodelling in response to dietary patterns and exercise training. Biology (Basel). 2022;11(6):882.
- Musale V, et al. Extracellular matrix remodelling in obesity and metabolic disease. Br Med Bull. 2023;147(1):84–101.
- Sun K, Li X, Scherer PE. Extracellular matrix remodelling in adipose tissue: overview and perspectives. Compr Physiol. 2023;13(1):1–35.
Autism, Neurodivergence and Hypermobility / EDS
- Baeza-Velasco C, et al. Autism in the context of joint hypermobility, hypermobility spectrum disorders and Ehlers–Danlos syndromes: systematic review and prevalence meta-analyses. Autism Res. 2025.
- Casanova EL, Sharp JL, Edelson SM, et al. The relationship between autism and Ehlers–Danlos syndromes/hypermobility spectrum disorders. J Pers Med. 2020;10(4):260.
- Cederlöf M, et al. Nationwide population-based cohort study of psychiatric disorders in individuals with Ehlers–Danlos syndrome or hypermobility syndrome and their siblings. BMC Psychiatry. 2016;16:207.
- Glans MR, et al. The relationship between generalised joint hypermobility, autism, ADHD and neurodevelopmental traits in adults. J Autism Dev Disord. 2022;52(9):3967–3978.
- Kindgren E, Quiñones Perez A, Knez R. Prevalence of ADHD and autism spectrum disorder in children with hypermobility spectrum disorders or hypermobile Ehlers–Danlos syndrome: a retrospective study. Neuropsychiatr Dis Treat. 2021;17:145–153.