Introduction
Despite increasing clinical recognition, lipoedema remains unusual among chronic diseases because no validated diagnostic biomarker or definitive imaging test exists. Lipoedema assessment continues to rely on pattern recognition and exclusion of differential diagnoses, which contributes to variation in reported prevalence and differences in clinical interpretation (1–3).
Attempts to establish objective diagnostic markers for lipoedema, including lymphoscintigraphy, MRI adipose mapping, ultrasound tissue evaluation and inflammatory biomarkers, have produced inconsistent findings.
There is disagreement as to whether lipoedema is a lymphatic disorder at all, and even the name lipoedema is contested. Some studies report normal lymphatic transport in early disease, while others identify secondary lymphatic dysfunction in more advanced stages. This variability has contributed to ongoing uncertainty in disease classification and reinforces the continued reliance on clinical diagnostic evaluation (4–6).
In response to this diagnostic uncertainty and following the review of current literature written below, a structured clinical assessment tool has been developed to support more consistent clinical evaluation in practice.
It is available for free download here:
Reframing Lipoedema: The 2020 Paradigm Shift & Its Implications
The 2020 international consensus document marked a shift in the conceptualisation of lipoedema. The condition was reframed as primarily a disorder of adipose tissue rather than one defined by lymphatic failure or oedema. The authors proposed that oedema observed in lipoedema should be understood as secondary to microvascular or inflammatory processes rather than as evidence of primary lymphatic pathology and proposed a name change to Lipalgia Syndrome. This position challenged earlier lymphology-derived models that had explained limb enlargement predominantly in oedema/fluid-based mechanisms (2,7).
This reclassification has not resolved uncertainty regarding the role of lymphatic dysfunction. The issue remains contested, particularly within European lymphology where combined lipolymphoedema models were, and continue to be, widely adopted (6,8). Differences in interpretation continue to influence clinical understanding, diagnostic practice, treatment approaches and research priorities.
Lymphatic Involvement: Primary, Secondary or Coexistant?
The extent and significance of lymphatic dysfunction in lipoedema remain areas of ongoing debate. Three broad positions are represented in the literature.
- The primary adipose disorder model, reflected in the 2020 consensus and subsequent guidance, proposes that lymphatic dysfunction is not a defining feature in early disease and, where present, is likely to be secondary. This interpretation places greater emphasis on adipose tissue mechanisms than on fluid accumulation (2,8).
- The spectrum model, commonly described as a lipolymphoedema continuum, suggests that progressive adipose expansion may result in secondary lymphatic overload and eventual impairment. Within this framework, lymphatic dysfunction is considered a potential later consequence of disease progression (6).
- A third perspective considers lymphoedema and lipoedema to be distinct conditions that may coexist due to shared risk factors, including obesity, hormonal influences and connective tissue susceptibility. In this model, coexistence does not imply a shared underlying mechanism (5).
Imaging studies have demonstrated both preserved and impaired lymphatic function in women with lipoedema. These findings support the view that the condition is heterogeneous rather than explained by a single underlying mechanism. Variation may reflect differences in disease stage, clinical presentation or limitations of current imaging techniques (4,9).
Diagnostic Challenges: Staging vs Phenotyping
Earlier classification systems for lipoedema relied mainly on morphological staging. These approaches focused on visible changes in limb contour and tissue texture. More recent consensus work has questioned the clinical usefulness of staging alone and identified several limitations.
Staging does not adequately reflect symptom burden, nor does it account for metabolic or inflammatory variability. It may also oversimplify the heterogeneity seen between women with the disease.
In response, newer frameworks have proposed classification based on observable disease expression including distribution pattern, symptom severity, functional impact and associated comorbidities. Such multidimensional models reflect broader developments in chronic disease medicine (3,7). Phenotype-based classification may better document individual variation and inform clinical decision-making.
Differential Diagnostic Overlap & Complexity
Lipoedema shares clinical features with several other conditions, which can make diagnostic differentiation challenging. These include lymphoedema, obesity, chronic venous disease, Dercum’s disease and connective tissue disorders.
Accurate diagnosis requires careful assessment of symmetry, pain characteristics, tissue texture, swelling behaviour and systemic features. Misclassification remains common and may lead to delayed diagnosis or inappropriate management (5,10). Precise differentiation is therefore important for treatment planning.
Implications for Clinical Practice
Changes in conceptual understanding have practical consequences for clinical care. Clinicians must recognise that current diagnostic frameworks are largely consensus-based rather than supported by validated objective criteria. Clinical judgement therefore remains central to assessment.
Thorough differential diagnosis is essential and multidisciplinary evaluation may be required in complex cases. Recent consensus documents increasingly emphasise functional outcomes, patient-reported symptoms and psychosocial impact, rather than reliance on morphological appearance alone (1,3). This reflects wider shifts in the management of chronic conditions.
Research Implications
The absence of objective diagnostic criteria represents a major barrier to progress in lipoedema research. It affects the accuracy of epidemiological estimates, the design of clinical trials, the evaluation of surgical outcomes and recognition within health policy frameworks.
Consensus documents have consistently identified priority research areas, including the development of validated diagnostic criteria, identification of biological markers, longitudinal studies of disease progression and phenotype-specific treatment strategies. These priorities reflect a transition from descriptive clinical observational studies towards investigation of underlying disease processes.
Pathophysiological Debate & Emerging Biological Models
Adipose Tissue Dysregulation
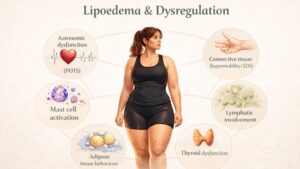
Despite improvements in clinical recognition, the biological mechanisms underlying lipoedema remain largely unknown. Contemporary literature increasingly describes the condition as a heterogeneous disorder of regional adipose tissue regulation. Proposed mechanisms involve interactions between adipocytes, microvascular structures, extracellular matrix components, lymphatic dynamics and endocrine signalling (11–13). This reflects the growing recognition that adipose tissue itself as an active metabolic and immunological organ.
Histopathological studies have demonstrated adipocyte hypertrophy, alterations in stromal architecture and evidence of chronic low-grade inflammatory infiltration in affected tissues. These findings support the hypothesis that lipoedema represents intrinsic dysregulation of adipose tissue biology rather than generalised metabolic adiposity (14). In contrast to obesity, adipose expansion in lipoedema typically follows characteristic regional distribution patterns, suggesting local susceptibility mediated by hormonal, vascular or connective tissue factors.
Microvascular Mechanisms
Microvascular dysfunction has long been proposed as a contributing mechanism. Observational studies have reported increased capillary fragility, altered vascular permeability and microangiopathic changes within affected adipose tissue (15). These alterations may contribute to interstitial fluid accumulation, mechanical stress and inflammatory signalling, although causality remains uncertain.
Inflammatory Processes
Chronic inflammatory processes have also been implicated. Adipose tissue analyses demonstrate macrophage infiltration and altered cytokine expression profiles consistent with inflammatory activation (14,16). Whether inflammatory signalling is a primary driver of adipose expansion or a secondary response to tissue hypertrophy remains unresolved.
Endocrine Involvement
Endocrine influences are suggested by clinical onset patterns. Many women report symptom development or progression during periods of hormonal fluctuation, including puberty, pregnancy and menopause. The 2020 international consensus highlighted the likely role of hormonal influences on adipose tissue, although the underlying mechanisms remain unclear (12).
Lymphatic Dysfunction
The role of lymphatic dysfunction continues to be debated. Imaging studies have demonstrated both preserved and impaired lymphatic transport in different disease stages (17). Current consensus documents state that primary lymphatic failure is not considered a defining feature of early lipoedema, although secondary lymphatic overload may occur with progressive adipose expansion (12,18). This distinction underpins ongoing discussion regarding whether lipolymphoedema represents a disease stage or a comorbidity.
Extracellular Matrix & Fascia
Alterations in extracellular matrix composition and fascial composition have emerged as additional areas of investigation. Changes in collagen organisation and tissue stiffness suggest that impaired mechanotransduction may influence adipocyte differentiation and regional tissue expansion (14). These observations align with broader mechanobiological models of adipose regulation.
Genetic Links
Familial clustering indicates a probable genetic link, although specific causation has not yet been identified. Proposed mechanisms include polygenic susceptibility affecting adipose metabolism, vascular integrity or connective tissue resilience (13). Associations with joint hypermobility and connective tissue disorders further support the hypothesis of structural tissue vulnerability contributing to disease expression.
Relationship with Obesity
The relationship between lipoedema and obesity remains complex. Although lipoedema adipose tissue often demonstrates relative resistance to calorie restriction, coexisting obesity may exacerbate inflammatory signalling, mechanical burden and metabolic risk (11). Contemporary consensus frameworks increasingly conceptualise lipoedema as a distinct adipose phenotype rather than a primary metabolic disorder, although partial pathophysiological overlap remains possible.
Research Priorities
Priority research areas continue to include clarification of biological markers, lymphatic involvement, improved phenotypic classification and longitudinal natural history studies (12,18).
Conclusion
This review was undertaken to examine the current literature on lipoedema in order to clarify areas of uncertainty, ongoing debate and inconsistency in clinical understanding. Despite increasing recognition of the condition, important questions remain regarding its underlying biology, classification and diagnostic boundaries. Variability in clinical presentation, overlap with related disorders and the absence of definitive objective markers continue to complicate both diagnosis and management.
By bringing together the available evidence, this review aims to provide clinicians with a coherent background to contemporary thinking on lipoedema assessment. In response to the diagnostic challenges identified, an evidence-based clinical assessment tool has been developed and is available to download below, to support more consistent evaluation in clinical practice.
Ongoing research will be essential to refine diagnostic frameworks, improve pathophysiological understanding and inform future approaches to classification and treatment. Until greater clarity is achieved, careful clinical judgement, supported by structured assessment, remains central to the management of lipoedema.
References
- Kruppa P, Georgiou I, Biermann N, Prantl L, Klein-Weigel P. Lipedema—pathogenesis, diagnosis and treatment options. Dtsch Arztebl Int. 2020;117:396–403.
- Bertsch T, Erbacher G, Elwell R. Lipoedema: a paradigm shift and consensus. J Wound Care. 2020;29(Suppl 11b):1–51.
- Herbst KL, Kahn LA, Iker E, Ehrlich C, Wright T, McHutchison L, et al. Standard of care for lipedema in the United States. Phlebology. 2021;36:779–796.
- Bilancini S, Lucchi M, Tucci S, Eleuteri P. Functional lymphatic alterations in patients suffering from lipedema. Angiology. 1995;46:333–339.
- Reich-Schupke S, Schmeller W, Brauer WJ, Cornely ME, Faerber G, Ludwig M, et al. S1 guidelines: Lipedema. J Dtsch Dermatol Ges. 2017;15:758–767.
- Faerber G, Bertsch T, Bickenbach J, et al. S2k guideline lipedema. J Dtsch Dermatol Ges. 2024;22:1303–1315.
- Wold LE, Hines EA, Allen EV. Lipedema of the legs: a syndrome characterised by fat legs and orthostatic oedema. Ann Intern Med. 1951;34:1243–1250.
- Schmeller W, Meier-Vollrath I. Lipolymphoedema: combined lipoedema and lymphoedema. Phlebologie. 2007;36:131–136.
- Rasmussen JC, Aldrich MB, Herbst KL, Tan IC, Zhu B, Darne C, et al. Lymphatic imaging in patients with lipedema. Lymphat Res Biol. 2017;15:1–8.
- Hansson E, Svensson H, Brorson H. Review of Dercum’s disease and proposal of diagnostic criteria, diagnostic methods, classification and management. Orphanet J Rare Dis. 2012;7:23.
- Al-Ghadban S, Cromer W, Allen M, Ussery C, Badowski M, Harris D, et al. Dilated blood and lymphatic microvessels, angiogenesis, increased macrophages, and adipocyte hypertrophy in lipedema thigh skin and fat tissue. J Obes. 2019;2019:8747461.
- Herbst KL. Rare adipose disorders masquerading as obesity. Acta Pharmacol Sin. 2012;33:155–172.
- Felmerer G, Stylianaki A, Hägerling R, Wang A, Ströbel P, Hollmén M, et al. Adipose tissue hypertrophy, lymphatic dysfunction, and collagen deposition in lipedema. Plast Reconstr Surg. 2020;145:1435–1443.
- Szolnoky G, Nemes A, Gavaller H, Forster T, Kemény L. Lipedema is associated with increased cutaneous capillary fragility. Lymphology. 2008;41:161–166.
- Rasmussen JC, Tan IC, Darne C, Zhu B, O’Donnell TF, Sevick-Muraca EM. Near-infrared fluorescence lymphatic imaging in lipedema. Lymphat Res Biol. 2021;19:1–9.
- Forner-Cordero I, Szolnoky G, Forner-Cordero A, Kemény L. Lipedema: an overview of its clinical manifestations, diagnosis and treatment. Clin Obes. 2012;2:86–95.
- Herbst KL. Subcutaneous adipose tissue diseases. Endotext. 2019.
- Faerber G, Bertsch T, Bickenbach J, et al. S2k guideline lipedema. J Dtsch Dermatol Ges. 2024;22:1303–1315.